A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the two steps in a nucleophilic addition mechanism?
A) Nucleophilic attack followed by protonation
B) Nucleophilic attack followed by deprotonation
C) Nucleophilic attack followed by substitution
D) Nucleophilic attack followed by elimination
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 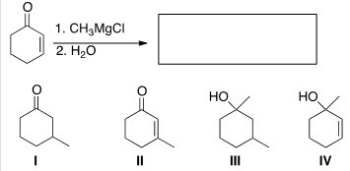
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 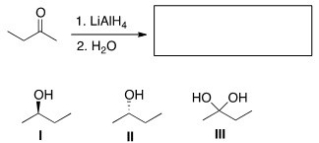
A) Only I
B) Only II
C) Only III
D) Only I and II
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 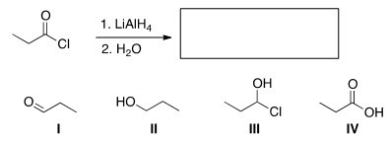
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the carbon-metal bond in the following organometallic reagents in order of decreasing polarity,starting with the most polar. 
A) II > I > III
B) II > III > I
C) III > I > II
D) III > II > I
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing reactivity in nucleophilic acyl substitution reactions,starting with the least reactive compound. 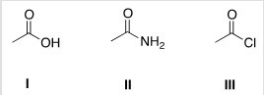
A) III < II < I
B) II < III < I
C) III < I < II
D) II < I < III
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing reagent in the reaction below? 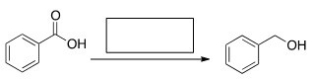
A) NaBH4/CH3OH
B) H2,Pd-C
C) LiAlH4 then H2O
D) MeMgBr then H2O
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product from the following reaction? 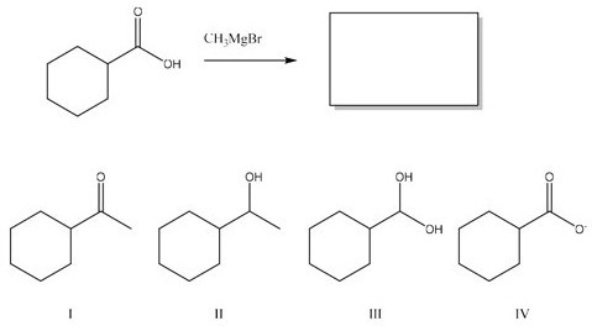
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A carbonyl group,C=O,and an alkene,C=C,double bonds are both sp2 hybridized.However,the chemistry of these two functional groups is very different.This can be explained by which of the following statements?
A) The bond angle of the carbonyl is larger than the bond angle of the alkene.
B) The electronegative oxygen of the C=O group makes this bond polar.
C) The bond of the C=C is longer that the bond of the C=O.
D) There is more steric crowding in the carbonyl than in the alkene.
F) A) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 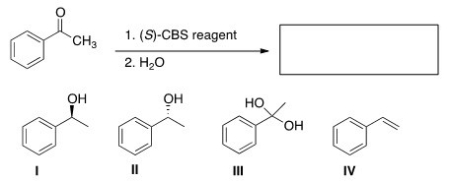
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
The behavior of acid chlorides is different from that of aldehydes in a reaction with a nucleophile because
A) the acid chloride contains a better leaving group
B) the aldehyde is more easily oxidized
C) the carbonyl of the aldehyde is more positive
D) the carbonyl of the aldehyde is less hindered
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the starting material has no stereogenic centers,when carbonyl compounds are reduced with a reagent such as LiAlH4 or NaBH4 and a new stereogenic center is formed,what will the composition of the product mixture be?
A) Forms a racemic mixture of the two possible enantiomers
B) Forms more of one enantiomer than another because of steric reactions around the carbonyl
C) Forms more of one enantiomer than another depending on the temperature of the reaction
D) Forms different products depending on the solvent used
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why are ketones less reactive than aldehydes?
A) Ketones are more sterically hindered
B) Ketones are less electron deficient due to donation from the two alkyl groups
C) The statement is false; ketones are more reactive than aldehydes
D) Both Ketones are more sterically hindered and Ketones are less electron deficient due to donation from the two alkyl groups
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 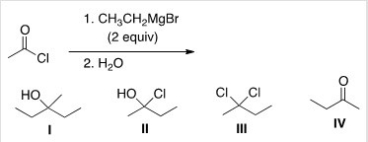
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What reagent can be used to cleave a silyl ether protecting group?
A) NaOMe
B) MeMgBr
C) Bu4NF
D) Pd/C
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which reagent can be used to reduce an acid chloride to an aldehyde?
A) NaBH4
B) LiAlH(OtBu) 3
C) LiAlH4
D) FeCl3
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 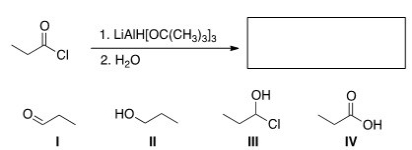
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What reagent would be used to reduce an amide to an amine?
A) NaBH4
B) LiAlH(OtBu) 3
C) LiAlH4
D) FeCl3
F) B) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 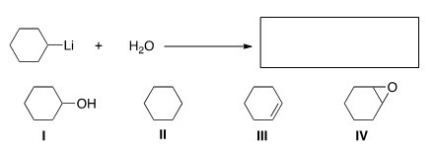
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 59
Related Exams